New Treatment for Allergic Response Targets Mast Cells
For Immediate Release
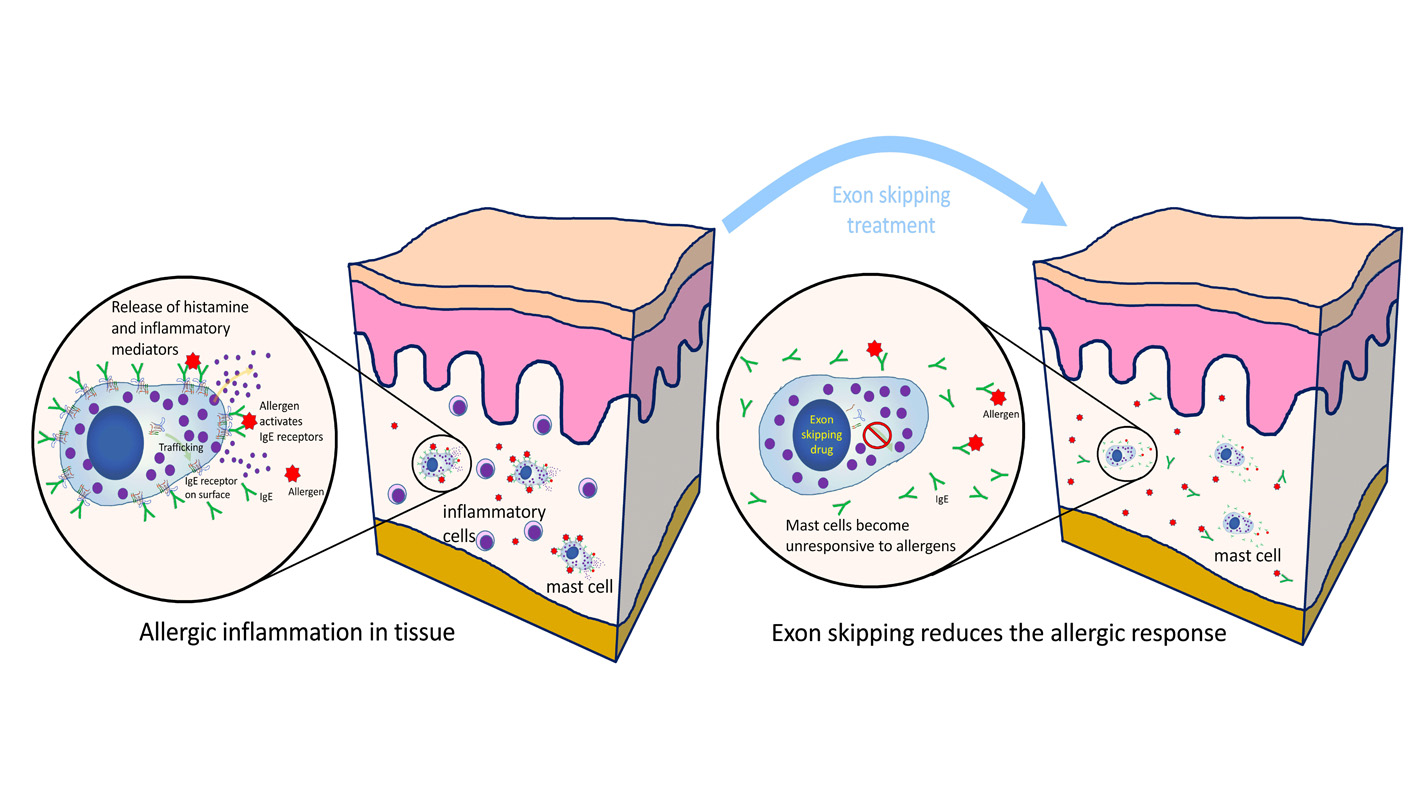
Researchers from North Carolina State University and the National Institutes of Health (NIH) have developed a method that stops allergic reactions by removing a key receptor from mast cells and basophils. Their work has implications for the treatment of skin allergies and asthma.
Allergic reactions are driven by mast cells and basophils – types of inflammatory cells found in tissues and the bloodstream, respectively, that function as part of our immune system. When you come into contact with an allergen – ragweed, for example – immunoglobulin E (IgE) specific to that allergen acts through its receptor on the mast cell, stimulating the mast cells and basophils to release mediators, such as histamine, that trigger an allergic response.
Currently, most allergy treatments focus on either stopping the effects of histamine and other mediators or on dampening the body’s overall immune response by use of steroids. Unfortunately neither approach is totally effective, and in the case of dampening the immune response, can have significant drawbacks.
Glenn Cruse, formerly of the NIH, current assistant professor of immunology at NC State and corresponding author of a paper describing the work, along with NIH colleague and co-author Dean Metcalfe, wanted to try and block the allergic reaction at its source.
Cruse and Metcalfe looked at a gene called MS4A2, which is only expressed in mast cells and basophils, and is responsible for forming the IgE receptor on the mast cell.
The researchers utilized a technique known as exon skipping, a form of RNA splicing, to eliminate the portion of one of the IgE receptor gene’s mRNA that is essential to making a protein which places the IgE receptor on the mast cell surface. The cell’s DNA remains unaffected. When the exon targeting therapy is stopped, the protein blocked by exon skipping is made again.
Cruse and Metcalfe tested their therapy on mast cells in vitro – where it eliminated activation of mast cells by allergen – and against allergic dermatitis in vivo, using a mouse model. Their results in vivo showed a marked reduction in the allergic dermatitis response in the mice.
“Asthma and allergic diseases affect up to 20 percent of people in developed countries, and their prevalence is increasing,” says Cruse. “By eliminating the expression of the IgE receptor on the surface of mast cells, we have identified an innovative and targeted approach with the potential to treat allergic inflammation in millions of patients worldwide.
“Due to the specificity of our approach for mast cells and basophils, it should have significant advantages over current therapies. However, it is important to note that while our findings are very promising, we are still in the early stages of developing this therapeutic approach.”
The research appears in Proceedings of the National Academy of Sciences. Funding was provided by the NIH Division of Intramural Research of NIAID and NHLBI.
-peake-
Note to editors: An abstract of the paper follows
“Exon skipping of FcεRIβ eliminates expression of the high affinity IgE receptor in mast cells with therapeutic potential for allergy”
DOI: 10.1073/pnas.1608520113
Authors: Glenn Cruse, North Carolina State University and National Institutes of Health; Tomoki Fukuyama, Greer K. Arthur, Wolfgang Bäumer, North Carolina State University; Yuzhi Yin, Avanti Desai, Michael A. Beaven and Dean D. Metcalfe, National Institutes of Health
Published: Proceedings of the National Academy of Sciences
Abstract:
Allergic diseases are driven by activation of mast cells and release of mediators in response to IgE-directed antigens. However, there are no drugs currently available that can specifically down-regulate mast cell function in vivo when chronically administered. Here, we describe an innovative approach for targeting mast cells in vitro and in vivo using antisense oligonucleotide-mediated exon skipping of the β-subunit of the high affinity IgE receptor (FcεRIβ) to eliminate surface high affinity IgE receptor (FcεRI) expression and function, rendering mast cells unresponsive to IgE-mediated activation. As FcεRIβ expression is restricted to mast cells and basophils, this approach would selectively target these cell types. Given the success of exon skipping in clinical trials to treat genetic diseases such as Duchenne muscular dystrophy, we propose that exon skipping of FcεRIβ is a potential approach for mast cell-specific treatment of allergic diseases.
- Categories:


