Birnessite Study Offers Insights For Electrochemistry, Energy Storage
For Immediate Release
The adsorption of ions from the electrolyte at an electrode surface is a ubiquitous process, of use for both existing and emerging electrochemical energy technologies. But what happens when these ions penetrate very small spaces? To address this question, researchers at NC State re-examined the behavior of a “classic” material, birnessite.
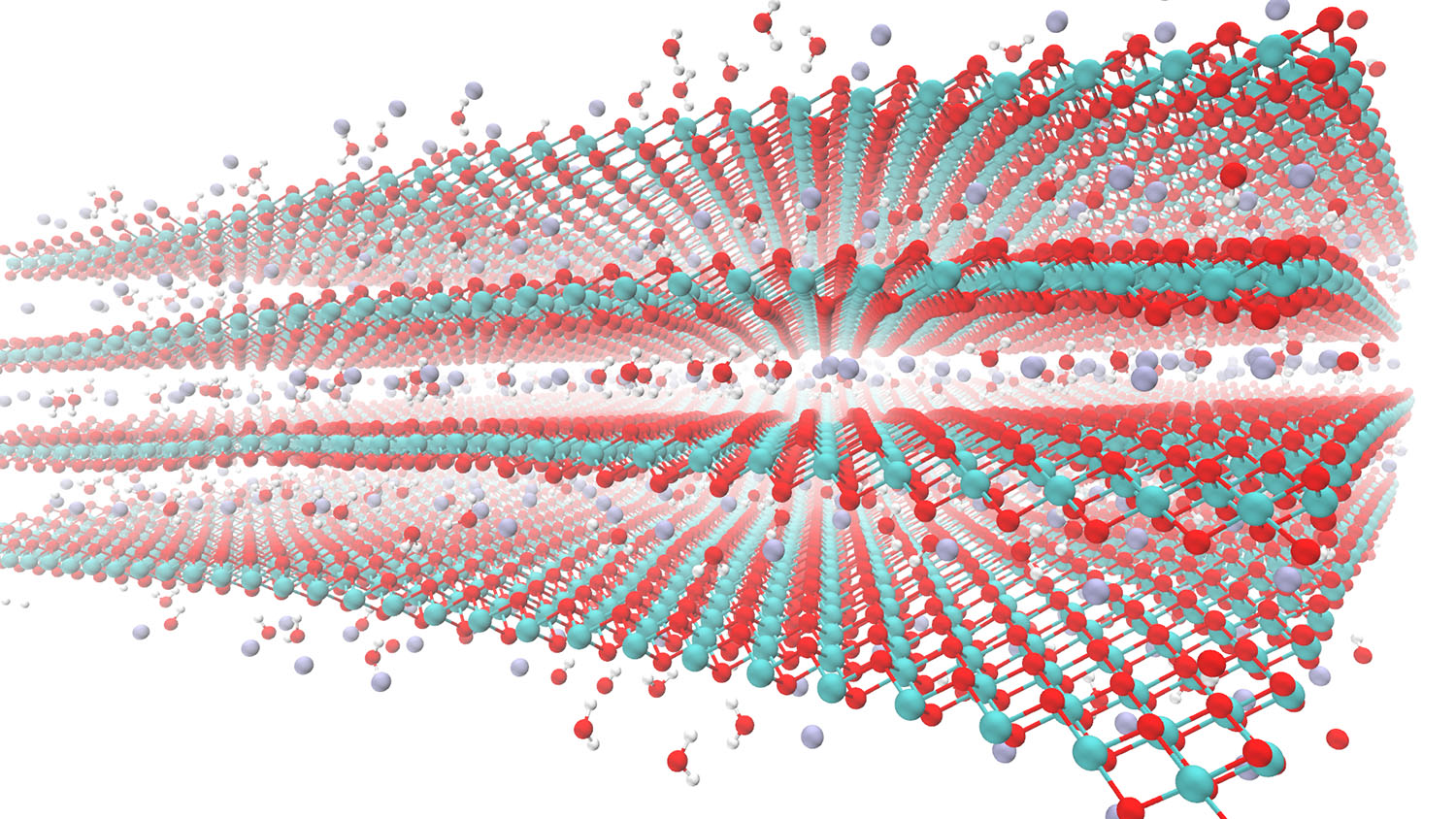
Birnessite is a hydrated layered form of manganese oxide that can quickly store and release a variety of positive ions from electrolytes for many cycles. This makes it promising for use in high-power electrochemical energy storage, or in emerging electrochemical technologies such as desalination and rare element recovery from water. What’s more, it’s an abundant material, easy to make, and non-toxic.
The mechanism by which birnessite can uptake and release cations has been described as both faradaic (involving charge-transfer) and non-faradaic (involving only electrostatic ion adsorption).
To address this debate, the researchers used both experimental and computational approaches.
“In the energy storage community, we normally think of charge storage as being either faradaic or non-faradaic,” says Shelby Boyd, first author of a paper on the work and a postdoctoral researcher at North Carolina State University. “At planar interfaces, faradaic refers to the specific adsorption of an ion to an electrode with corresponding charge transfer, as in a redox reaction. Non-faradaic refers to purely electrostatic adsorption without charge transfer. People have largely presented these mechanisms of charge storage as being mutually exclusive. But what we found with birnessite is that the nanoconfined interlayer structural water mitigates the interactions between intercalated cation and the birnessite. This results in an intermediate behavior from the two types of adsorption extremes at planar interfaces.”
The researchers were also able to prove experimentally and theoretically that water in between the layers of birnessite is effectively serving as a buffer that makes capacitive behavior possible without causing significant structural change in the birnessite.
Ultimately, the researchers say the findings highlight two future directions for the work, both of which are promising for the broader field of electrochemistry.
“The field of electrochemistry is undergoing a renaissance,” says Veronica Augustyn, corresponding author of the paper and an assistant professor of materials science and engineering at NC State. “The ability to connect experimental results with atomistic-scale modeling of the electrochemical interface allows us to probe deeper than ever before and ask questions like: What roles are the solvent playing? What might happen when the reaction occurs under confinement? By understanding the capacitive mechanism of a material like birnessite, we set the stage for understanding more complex electrochemical reactions.”
The paper, “Effects of Interlayer Confinement and Hydration on Capacitive Charge Storage in Birnessite,” is forthcoming from Nature Materials. The paper was co-authored by Saeed Saeed, a Ph.D. student at NC State; Karthik Ganeshan and Adri C.T. van Duin of Pennsylvania State University; Wan-Yu Tsai and Nina Balke of Oak Ridge National Laboratory; and Tao Wu and De-en Jiang of University of California, Riverside. Balke will join the NC State faculty as an associate professor of materials science and engineering in August 2021.
This work was supported as part of the Fluid Interface Reactions, Structures and Transport, an Energy Frontier Research Center funded by the U.S. Department of Energy, Office of Science, Basic Energy Sciences at Oak Ridge National Laboratory. Boyd was also supported by the National Science Foundation Graduate Research Fellowship Program (GRFP).
-shipman-
Note to Editors: The study abstract follows.
“Effects of Interlayer Confinement and Hydration on Capacitive Charge Storage in Birnessite”
Authors: Shelby Boyd, Saeed Saeed and Veronica Augustyn, North Carolina State University; Karthik Ganeshan and Adri C.T. van Duin, Pennsylvania State University; Wan-Yu Tsai and Nina Balke, Oak Ridge National Laboratory; and Tao Wu and De-en Jiang, University of California, Riverside
Published: August 2, Nature Materials
DOI: 10.1038/s41563-021-01066-4
Abstract: Nanostructured birnessite exhibits high specific capacitance and nearly ideal capacitive behaviour in aqueous electrolytes, rendering it an important electrode material for low-cost, high-power energy storage devices. The mechanism of electrochemical capacitance in birnessite has been described as both Faradaic (involving redox) and non-Faradaic (involving only electrostatic interactions). To clarify the capacitive mechanism, we characterized birnessite’s response to applied potential using ex situ X-ray diffraction, electrochemical quartz crystal microbalance, in situ Raman spectroscopy and operando atomic force microscope dilatometry to provide a holistic understanding of its structural, gravimetric and mechanical responses. These observations are supported by atomic-scale simulations using density functional theory for the cation-intercalated structure of birnessite, ReaxFF reactive force field-based molecular dynamics and ReaxFF-based grand canonical Monte Carlo simulations on the dynamics at the birnessite–water–electrolyte interface. We show that capacitive charge storage in birnessite is governed by interlayer cation intercalation. We conclude that the intercalation appears capacitive due to the presence of nanoconfined interlayer structural water, which mediates the interaction between the intercalated cation and the birnessite host and leads to minimal structural changes.


