Bio-Inspired ‘Nano-Cocoons’ Offer Targeted Drug Delivery Against Cancer Cells
For Immediate Release
Biomedical engineering researchers have developed a drug delivery system consisting of nanoscale “cocoons” made of DNA that target cancer cells and trick the cells into absorbing the cocoon before unleashing anticancer drugs. The work was done by researchers at North Carolina State University and the University of North Carolina at Chapel Hill.
“This drug delivery system is DNA-based, which means it is biocompatible and less toxic to patients than systems that use synthetic materials,” says Dr. Zhen Gu, senior author of a paper on the work and an assistant professor in the joint biomedical engineering program at NC State and UNC Chapel Hill.
“This technique also specifically targets cancer cells, can carry a large drug load and releases the drugs very quickly once inside the cancer cell,” Gu says.
“In addition, because we used self-assembling DNA techniques, it is relatively easy to manufacture,” says Wujin Sun, lead author of the paper and a Ph.D. student in Gu’s lab.
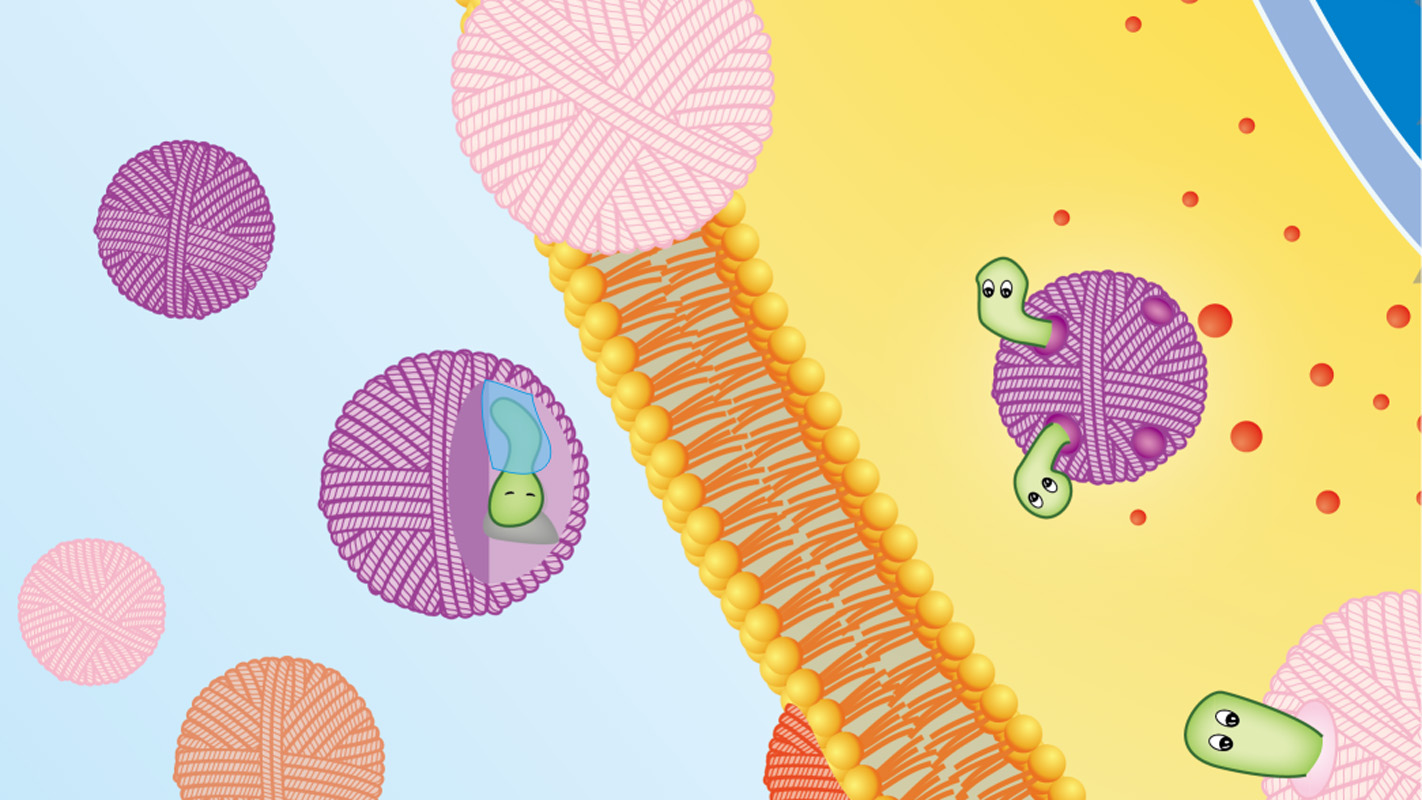
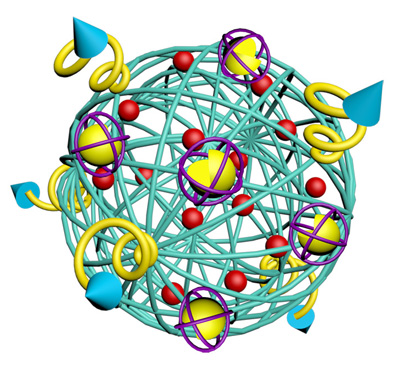
Each nano-cocoon is made of a single strand of DNA that self-assembles into what looks like a cocoon, or ball of yarn, that measures 150 nanometers across.
The core of the nano-cocoon contains the anticancer drug doxorubicin (DOX) and a protein called DNase. The DNase, an enzyme that would normally cut up the DNA cocoon, is coated in a thin polymer that traps the DNase like a sword in a sheath.
The surface of the nano-cocoon is studded with folic acid ligands. When the nano-cocoon encounters a cancer cell, the ligands bind the nano-cocoon to receptors on the surface of the cell – causing the cell to suck in the nano-cocoon.
Once inside the cancer cell, the cell’s acidic environment destroys the polymer sheath containing the DNase. Freed from its sheath, the DNase rapidly slices through the DNA cocoon, spilling DOX into the cancer cell and killing it.
“We’re preparing to launch preclinical testing now,” Gu says. “We’re very excited about this system and think it holds promise for delivering a variety of drugs targeting cancer and other diseases.”
The paper, “Cocoon-Like Self-Degradable DNA-Nanoclew for Anticancer Drug Delivery,” was published online Oct. 13 in the Journal of the American Chemical Society. Co-authors include Yue Lu, a Ph.D. student in Gu’s lab; Margaret Reiff, an undergraduate student in the joint biomedical engineering department; Tianyue Jiang, a Ph.D. student in the joint biomedical engineering department and at the China Pharmaceutical University; and Dr. Ran Mo, a former postdoctoral researcher in the joint biomedical engineering department now at the China Pharmaceutical University.
This research was supported by the North Carolina Translational and Clinical Sciences Institute under grant number 1UL1TR001111 and with funding from NC State and UNC Chapel Hill.
-shipman-
Note to Editors: The study abstract follows.
“Cocoon-Like Self-Degradable DNA-Nanoclew for Anticancer Drug Delivery”
Authors: Wujin Sun, Yue Lu, Margaret Reiff, and Zhen Gu, North Carolina State University and University of North Carolina at Chapel Hill; Ran Mo and Tianyue Jiang, North Carolina State University, University of North Carolina at Chapel Hill, and China Pharmaceutical University
Published: Oct. 13, 2014, Journal of the American Chemical Society
DOI: 10.1021/ja5088024
Abstract: A bio-inspired cocoon-like anticancer drug delivery system consisting of a deoxyribonuclease (DNAse)-degradable DNA nanoclew (NCl) embedded with acid-responsive DNAse I nanocapsule (NCa) was developed for targeted cancer treatment. The NCl was assembled from a long chain single stranded DNA synthesized by the rolling circle amplification (RCA). Multiple GC-pair sequences were integrated in the NCl for enhanced loading capacity of anticancer doxorubicin (DOX). Meanwhile, negatively charged DNAse I was encapsulated in a positively charged acid-degradable polymeric nanogel to facilitate decoration of DNAse I into NCl by electrostatic interaction. At an acidic environment, the activity of DNAse I was activated due to the acid-triggered shedding of the polymeric shell of NCa, resulting in the cocoon-like self-degradation of NCl and promoting the release of DOX for enhanced therapeutic efficacy.
- Categories:


