For Immediate Release
Researchers at North Carolina State University and the University of North Carolina at Chapel Hill have developed a new drug delivery technique that uses a biodegradable liquid metal to target cancer cells. The liquid metal drug delivery method promises to boost the effect of cancer drugs. To date, the technique has only been tested in an animal model.
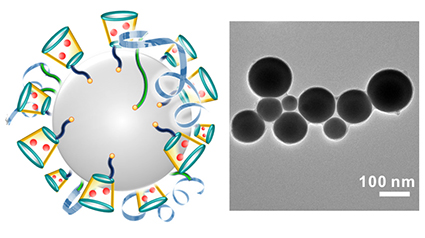
“The advance here is that we have a drug-delivery technique that may enhance the effectiveness of the drugs being delivered, can help doctors locate tumors, can be produced in bulk, and appears to be wholly biodegradable with very low toxicity,” says Zhen Gu, corresponding author of a Nature Communications paper on the work and an assistant professor in the joint biomedical engineering program at NC State and UNC-CH. “And one of the advantages of this technique is that these liquid metal drug carriers – or ‘nano-terminators’ – are very easy to make.”
To create the nano-terminators, researchers place the bulk liquid metal (gallium indium alloy) into a solution that contains two types of molecules called polymeric ligands. The solution is then hit with ultrasound, which forces the liquid metal to burst into nanoscale droplets approximately 100 nanometers in diameter. The ligands in the solution attach to the surface of the droplets as they break away from the bulk liquid metal. Meanwhile, an oxidized “skin” forms on the surface of the nanodroplets. The oxidized skin, together with the ligands, prevents the nanodroplets from fusing back together.
The anticancer drug doxorubicin (Dox) is then introduced into the solution. One of the ligands on the nanodroplet sucks up the Dox and holds on to it. These drug-laden nanodroplets can then be separated from the solution and introduced into the bloodstream.
The second type of ligand on the nanodroplets effectively seeks out cancer cells, causing receptors on the surface of the cancer cell to latch on to the nanodroplets. The cancer cell then absorbs the nanodroplets.

Once absorbed, the higher level of acidity inside the cancer cell dissolves the oxidized skin of the nanodroplets. This releases the ligands, which will go on to release the Dox inside the cell.
“Without the oxidized skin and ligands, the nanodroplets fuse together, forming larger drops of liquid metal,” says Michael Dickey, a co-author on this paper and professor in the Department of Chemical and Biomolecular Engineering at NC State. “These larger droplets are fairly easy to detect using diagnostic techniques, which can potentially help doctors locate tumors.”
Meanwhile, the liquid metal continues reacting with the acidic environment in the cancer cell and dissolves, releasing gallium ions. Interestingly, these gallium ions enhance the performance of anticancer drugs – including their effectiveness against drug-resistant cell lines.
In addition, this process gradually degrades the metal, minimizing long-term toxicity.
“Based on in vitro tests, we believe the liquid metal degrades completely in a matter of days into a form that the body can successfully absorb or filter out, without notable toxic effects,” says Yue Lu, a Ph.D. student in Gu’s lab.
The researchers have tested the liquid metal technique in a mouse model, and found that it is significantly more effective than Dox alone at inhibiting the growth of ovarian cancer tumors. Importantly, the researchers tracked the mice for up to 90 days, and found no signs of toxicity related to the liquid metal.
“This was a proof-of-concept study, but very encouraging,” Gu says. “Like the fictional Terminator, this carrier is transformable: smashed from bulk material, fused inside cancer cells and eventually degraded and cleared. We are hoping to do additional testing in a large animal study to get closer to potential clinical trials.”
The paper, “Transformable liquid-metal nanomedicine,” is published Dec. 2 in Nature Communications, and is open access. The paper was co-authored by Quanyin Hu, Dennis Pacardo, Chao Wang, Wujin Sun, and Frances Ligler of the joint biomedical engineering program; and by Michael Dickey and Yiliang Lin of NC State’s Department of Chemical and Biomolecular Engineering. The work was supported by NC TraCS, NIH’s Clinical and Translational Science Awards at UNC-CH, under grant number 1UL1TR001111; and by the National Science Foundation under grant number CMMI-0954321.
-shipman-
Note to Editors: The study abstract follows.
“Transformable liquid-metal nanomedicine”
Authors: Yue Lu, Quanyin Hu, Dennis B. Pacardo, Chao Wang, Wujin Sun, Frances S. Ligler, and Zhen Gu, North Carolina State University and the University of North Carolina at Chapel Hill; Yiliang Lin and Michael D. Dickey, North Carolina State University
Published: Dec. 2, Nature Communications
DOI: 10.1038/NCOMMS10066
Abstract: To date, numerous inorganic nanocarriers have been explored for drug delivery systems (DDSs). However, the clinical application of inorganic formulations has often been hindered by their toxicity and failure to biodegrade. We describe here a transformable liquid-metal nanomedicine, based on a core–shell nanosphere composed of a liquid-phase eutectic gallium-indium core and a thiolated polymeric shell. This formulation can be simply produced through a sonication-mediated method with bioconjugation flexibility. The resulting nanoparticles loaded with doxorubicin (Dox) have an average diameter of 107 nm and demonstrate the capability to fuse and subsequently degrade under a mildly acidic condition, which facilitates release of Dox in acidic endosomes after cellular internalization. Equipped with hyaluronic acid, a tumour-targeting ligand, this formulation displays enhanced chemotherapeutic inhibition towards the xenograft tumour-bearing mice. This metal-based DDS with fusible and degradable behaviour under physiological conditions provides a new strategy for engineering theranostic agents with low toxicity.
- Categories:



